This week: Consider this your formal advance apology.
I am a biochemist and molecular biologist by trade, and I have a bad habit of looking at the things I love and asking why they work. Whisky, watches, and cars all have extraordinary science underneath them, and this newsletter exists because I could not stop thinking about it.
This is the first of at least six science issues planned for the year. It takes about as long to put together as an actual research paper, except this one will not put you to sleep. My published work, on the other hand, absolutely will. Link here if you need it.
No drink suggestion this time around. Trust me, it will only make the chemistry harder to follow (although…. I can’t confirm or deny I’ve never had drinks in chem class. I did take a beer brewing science class, though). Let's get into it.
Whisk(e)y
POURED — The Wood Whisperer: Inside the Chemistry of Cask Maturation

One of Springbank’s dunnage warehouses from my April 2024 trip.
When a distiller fills a cask and rolls it into a warehouse, they are not simply storing spirit. They are initiating a slow-motion chemical experiment that will run for years, sometimes (hopefully!) decades, with no do-overs. The barrels themselves are the laboratory, the reagents, and the reactor all at once. Quite a few distillers believe that up to 60-80% of the flavor components in a finished Scotch malt whisky come from barrel maturation and the oak wood used [1], which is a staggering number when you consider that the spirit arrives at the cask completely colorless and barely resembling what it will eventually become. Understanding why requires getting into the wood itself, literally.
Oak is not a passive container. At the structural level, oak wood consists of cellulose (38-52%), hemicellulose (25-30%), lignin (22-25%), and extractives that make up the remaining fraction [2]. Think of cellulose as the load-bearing skeleton; it gives the wood its physical strength and contributes almost nothing to flavor. Hemicellulose (a shorter, branched sugar polymer, picture it as scaffolding wrapped around the cellulose fibers) is more reactive: in the presence of ethanol, it gives up its component sugars, which carry sweetness and contribute to the mouthfeel and roundness you associate with a well-aged dram [3]. Then there is lignin, the compound that holds everything together and is responsible for some of the most prized flavor molecules in the glass. Lignin (a complex, three-dimensional organic polymer that gives wood its rigidity, think of it as the structural glue between wood cells), when broken down by alcohol exposure, yields vanillin [4], the same molecule that defines the smell of a vanilla bean, along with guaiacol (a smoky, phenolic compound), oak lactones (cyclic ester molecules that produce coconut and woody aromas), and a cascade of other aromatics that form the backbone of barrel character.
You all still doing alright? I find stepping away, and staring into the void to process information if my head hurts. I stare off at work a lot, this lets me process what I read.
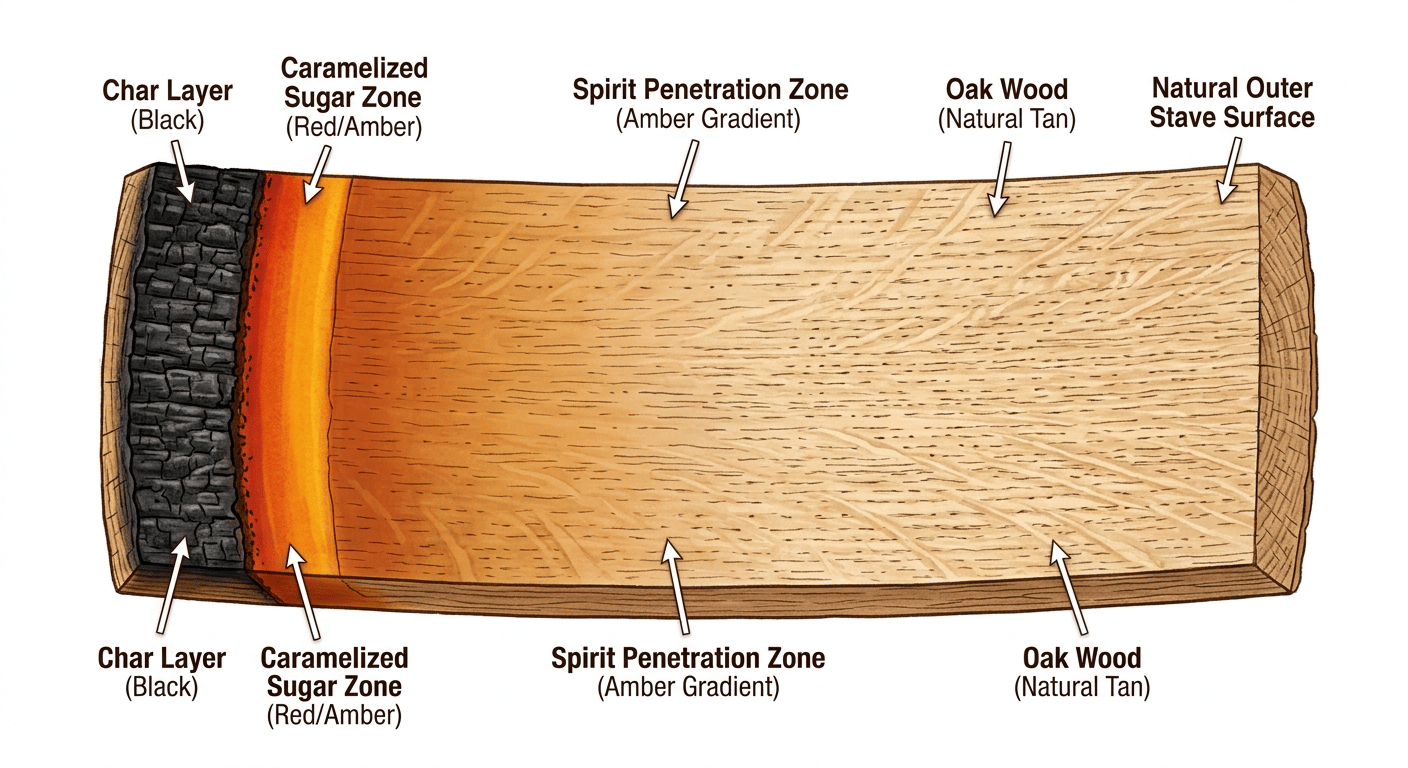
Think of this as you go left to right, each layer is behind the previous. So char layer →sugar zone → spirit penetration zone, etc...
The cask breathes, and that breathing matters as much as the wood chemistry. In summer, the contents of a barrel expand and some air and alcohol vapor is exhaled from the cask. As temperatures cool and the contents contract, the cask inhales fresh air, contributing to the loss of about 2% of spirit in Scotland each year, the so-called Angels' Share [3]. That oxygen exchange is not waste; it is chemistry. Oxidation (a reaction in which molecules lose electrons, here converting ethanol into new flavor-active compounds) runs continuously inside the cask: oxygen converts ethanol into carboxylic acids, which then react back with ethanol to produce esters (fragrant, fruity molecules) with floral overtones. Tannins (polyphenolic compounds extracted from wood that create the dry, mouth-coating sensation you feel) act as natural oxidizing agents themselves, removing sulfurous off-notes from the raw spirit and driving color development [3]. The whisky is not aging passively. It is reacting.
The final variable is cask history, and it determines the intensity of everything above. A first-fill ex-bourbon barrel floods incoming spirit with extraction compounds quickly. A third-fill cask is largely spent on wood extraction but may carry residual wine compounds absorbed into the grain [5]. This is why distillers carefully track fill numbers; it is not superstition, it is inventory management of a slow chemical reaction. It also explains why the debate around older whisky always being better whisky is flawed at its core: age is a proxy for wood contact time, not quality. A spirit spending 25 years in an exhausted cask may be far less interesting than one pulled at 12 from a first-fill barrel at the peak of its development. The wood does the work. Time is just how long you let it run.

Natural color, 32yo cask strength Springbank straight from the barrel! From my April 2024 trip.
Watches
WORN — The Dirty Secret in the Cage: What the Tourbillon Actually Does (and Doesn't Do)

Grail watch for me… Audemars Piguet Tourbillion. ~$200,000.
Before we get into the argument, a quick definition for everyone who just asked "the what?" A tourbillon (French for "whirlwind," pronounced tour-BEE-yohn) is a watchmaking complication, meaning a function beyond simply displaying hours and minutes, in which the most delicate timekeeping components of a mechanical watch are mounted inside a small rotating cage that spins continuously, usually once per minute. You can see it moving on the dial. It is mesmerizing, it is outrageously complex to build, and it typically adds tens of thousands of dollars to the price of a watch. It was also, as this section will argue, made functionally obsolete roughly a hundred years ago, and the industry has been very quietly not talking about that ever since.
The tourbillon was patented by Abraham-Louis Breguet in 1801, conceived to solve a real and documented problem in pocket watches. The escapement (the mechanism that regulates the release of energy from the mainspring to the gear train, the part that makes a watch tick at a controlled rate) and the hairspring (a tiny, extraordinarily fine coiled spring that drives the back-and-forth oscillation of the balance wheel, the heartbeat of the watch) are both exquisitely sensitive to gravitational pull. When a pocket watch sat in one position for hours, upright in a waistcoat pocket, gravity pulled on the hairspring unevenly, causing the escapement to run slightly fast or slow depending on orientation [6]. Breguet's solution was elegant: mount thse entire regulating organ in a cage and rotate it continuously. If gravity pulls from all directions equally over time, its errors average out. In a pocket watch, worn vertically for most of its life, this worked. Properly executed, a tourbillon genuinely improved accuracy. Problem solved, for about a century [7].

Then the wristwatch arrived, and the physics collapsed. A wristwatch is never still. It is vertical, horizontal, upside down, shaking hands, typing, waving down a cab. Gravity attacks from everywhere, all the time. In this environment, a tourbillon loses its advantage, because the watch is already averaging its own positional errors through constant random motion [7]. Modern escapement engineering compounds this further: silicon (a hard, non-metallic element used in modern hairsprings because it is non-magnetic, lightweight, and immune to temperature fluctuations) hairsprings, anti-magnetic alloys, and advanced free-sprung balance wheels have largely neutralized the positional errors Breguet was fighting. Many non-tourbillon movements are demonstrably more precise than tourbillons [7]. The problem the tourbillon solved no longer needs solving, at least not the way Breguet solved it.
So why does nearly every prestige manufacture still build them, and why does the market keep rewarding them handsomely? Because the tourbillon became something else entirely. Today it exists as proof of mastery, a complication that serves no commercial purpose and offers no shortcuts [7]. A traditional cage tourbillon contains upward of 70 hand-finished components weighing as little as 0.3 grams total. Building one under a microscope with a pair of tweezers is not watchmaking; it is surgery. The industry has also pushed the concept toward engineering that addresses the wristwatch problem more honestly: the new Jaeger-LeCoultre Master Hybris Inventiva Gyrotourbillon features a patented triple-axis tourbillon covering 98% of possible positional orientations [8], attempting to compensate for gravity across multiple planes simultaneously. Whether that final coverage percentage meaningfully improves timekeeping on a wrist is debatable. Whether the engineering required to make three nested cages rotate on intersecting axes inside a watch case is genuinely extraordinary is not debatable at all. The honest version of the tourbillon story is this: it was a brilliant solution to a real problem that no longer exists in the form it was designed for, and the industry kept building it because the thing is simply too beautiful to stop.

Jaeger Lecoultre (JLC) Duometre Spherotourbillon
Still good?
Wheels
DRIVEN — Corn in the Tank: The Real Science Behind E15

Something is changing at the gas pump this summer, and most drivers passing the new "Unleaded 88" label on the dispenser have no idea what it means or why it is there. The EPA issued an emergency fuel waiver effective May 1, 2026 to allow nationwide sales of E15, gasoline blended with 15% ethanol, framing it as fortifying the domestic fuel supply ahead of the summer driving season [9]. The waiver arrived against a specific backdrop: rising fuel prices driven by disruption to oil shipments through the Strait of Hormuz [10]. The policy decision is wrapped in economics and geopolitics, but underneath all of that is a chemistry story, and the chemistry is worth understanding clearly before you decide whether Unleaded 88 belongs in your tank.
Ethanol (grain alcohol, the same molecule that gives bourbon its backbone, produced by fermenting sugars from grain, in this case primarily corn) has been blended into American gasoline for decades. The standard blend, E10, has been in the fuel supply long enough that most drivers have never known anything different. The move to E15 is, in one sense, a small numerical step, five more percentage points of alcohol. In another sense it is a meaningful chemistry shift, because ethanol and gasoline are fundamentally different types of molecules. Gasoline is a dense mixture of hydrocarbons (molecules built entirely from hydrogen and carbon atoms, which burn with extremely high energy release). Ethanol, by contrast, contains an oxygen atom in its molecular structure, and that oxygen atom is why it carries less energy. The energy content of ethanol is approximately 76,100 BTU (British Thermal Unit, a measure of heat energy) per gallon, compared to roughly 114,100 BTU per gallon for pure gasoline [11], a gap of about 33%. The same corn fermentation chemistry that fills a bourbon barrel is, essentially, diluting the energy density of your fuel. Moving from E10 to E15 narrows that gap only slightly, but the EPA estimates most cars will see a 1.5-2% drop in fuel economy as a result [12].
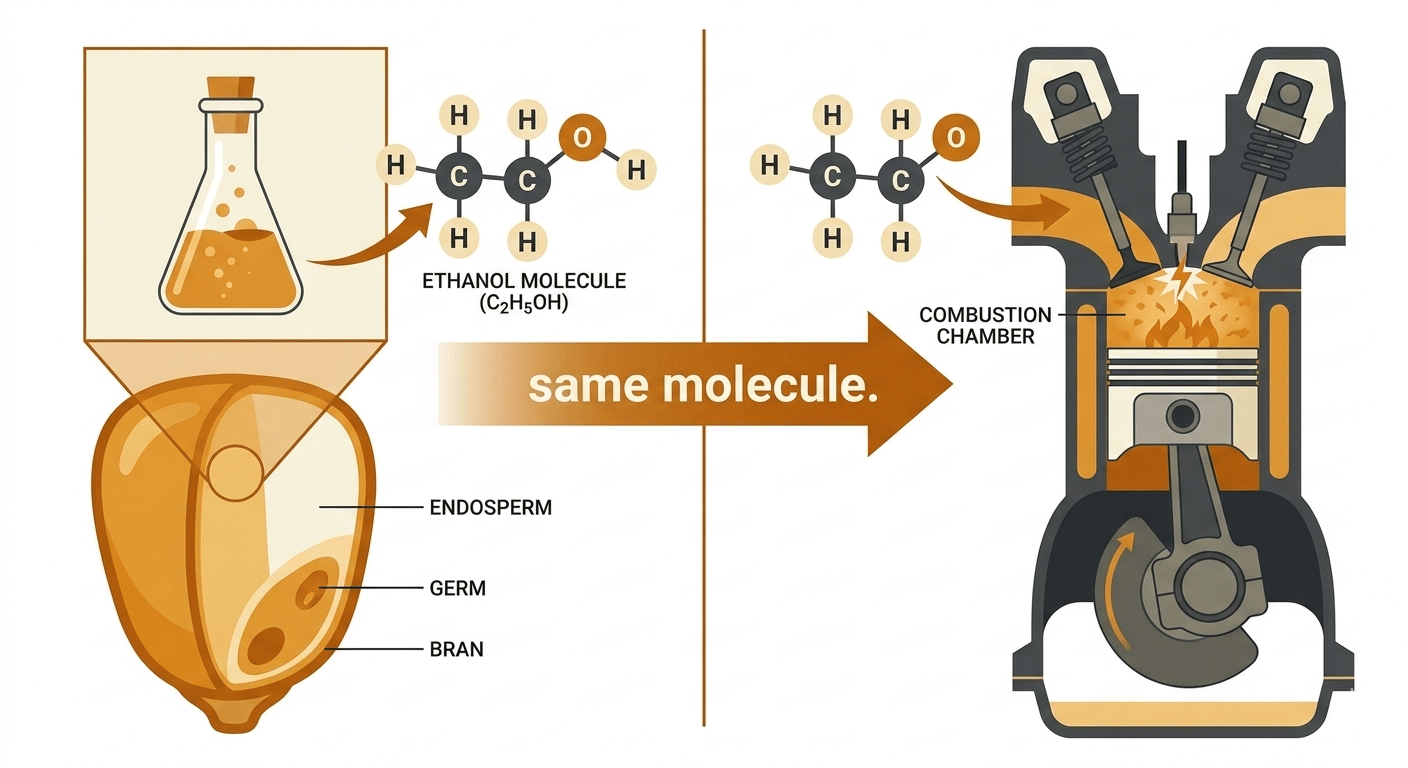
The octane story partially compensates, and this is where the chemistry gets interesting for anyone running a performance engine. Ethanol has a Research Octane Number (RON, a standardized measure of a fuel's resistance to premature detonation, or knock) of approximately 109 [13], significantly higher than standard 87 octane pump gas. Higher octane allows an engine with sufficient compression ratio to extract more work from each combustion cycle, partially offsetting the energy density penalty, which is precisely why E15 is marketed as Unleaded 88. Bourbon distillers have known for generations that corn fermentation produces a high-proof, energetic alcohol; the irony is that the same molecular properties that make corn ethanol punchy in a glass make it a slightly weaker fuel in a cylinder. For a stock modern car, the net effect is a wash that slightly favors your wallet at the pump, since E15 typically runs about 25 cents per gallon cheaper [12]. For a modified car running a tune calibrated to a specific fuel map and air-fuel ratio, the shift in ethanol percentage matters more: a few extra percent of alcohol can lean out a tune enough to affect power delivery and, in a worst case, engine safety margins.
The concern that gets less coverage than the pump price story is what ethanol does to materials inside older fuel systems. Ethanol is a polar solvent (a molecule that, due to its oxygen atom, carries a slight electrical charge, allowing it to dissolve materials that pure gasoline cannot) and is hygroscopic (meaning it actively pulls moisture out of the surrounding air). In older vehicles and small engines, anything with a carburetor, rubber fuel lines not engineered for alcohol exposure, or aluminum components in the fuel delivery system, higher ethanol content can cause seals to deteriorate, swell, or shrink, and can deposit water-carried contaminants inside injectors and carburetors [14]. Modern vehicles built after 2001 use fuel system components engineered to tolerate blends well above E15, so the risk is low for the vast majority of cars on the road. But if you own a boat, a motorcycle, a classic car, or anything with a small carbureted engine, E15 is not your fuel. And that is worth knowing before the pump changes without announcement.
Final Thoughts: This really shouldn’t harm your car, but be prepared for less gas mileage.
Outro: Well, that was a lot wasn't it? Hopefully no bad flashbacks from high school chemistry class! We covered wood chemistry, a spinning cage that technically should not exist anymore, and corn with a second job. Not bad for a Wednesday.
I have comments on the website, so pretty please, with sugar on top, let me know what you think! This one was genuinely fun to research, and if you made it all the way here without Googling "what is a tannin," I am proud of you. Got ideas for the next issue? A topic that deserves the deep dive treatment? I have comments turned on for the website, so let me know and I will see what I can extract from the barrel.
Have a great rest of the week. Drive safe, watch your octane, and maybe pour something worth thinking about tonight.
Poured. Worn. Driven.
Wristmas & The W’s
References:
POURED
[1] Whiskipedia, "The Science of Whisky Maturation." https://whiskipedia.com/fundamentals/whisky-science/
[2] Whisky Science Blog, "Oaky Flavours." http://whiskyscience.blogspot.com/2011/02/oaky-flavours.html
[3] Azu Spirits, "Maturation in Scotch Whisky." https://www.azuspirits.co.uk/blog/maturation
[4] The Glenlivet, "Whisky Maturation Process: Cask Maturation." https://www.theglenlivet.com/en/our-community/articles/whisky-maturation-process/
[5] Global Whiskey Authority, "Cask Types and Whiskey Maturation." https://globalwhiskeyauthority.com/cask-types-and-whiskey-maturation
WORN
[6] Wikipedia, "Tourbillon." https://en.wikipedia.org/wiki/Tourbillon
[7] Outlook Luxe, "The Tourbillon Explained: From Accuracy Myth to Modern Mastery." https://luxe.outlookindia.com/watches-jewellery/watches/the-tourbillon-explained-from-accuracy-myth-to-modern-mastery
[8] Jaeger-LeCoultre, "Watches and Wonders 2026." https://www.jaeger-lecoultre.com/us-en/news/watchmaking/watches-and-wonders-2026
DRIVEN
[9] U.S. EPA Press Release, "EPA Fortifies Domestic Fuel Supply, Provides Americans with Relief at the Pump by Approving Nationwide E15." March 25, 2026. https://www.epa.gov/newsreleases/epa-fortifies-domestic-fuel-supply-provides-americans-relief-pump-approving-nationwide
[10] Winston & Strawn, "EPA Issues New Emergency E15 Waiver and Renewable Fuel Standard Rule for 2026-2027." April 2026. https://www.winston.com/en/blogs-and-podcasts/winston-and-the-legal-environment/epa-issues-new-emergency-e15-waiver-and-renewable-fuel-standard-rule-for-2026-2027-amid-rising-fuel-costs-associated-with-iran-war
[11] Wikipedia, "Gasoline Gallon Equivalent." https://en.wikipedia.org/wiki/Gasoline_gallon_equivalent
[12] The Drive, "The Feds Plan to Start Diluting Gasoline This May: Explained." March 2026. https://www.thedrive.com/news/the-feds-plan-to-start-diluting-gasoline-this-may-explained
[13] Wikipedia, "Alcohol Fuel." https://en.wikipedia.org/wiki/Alcohol_fuel
[14] Penn State Extension, "Fuel Ethanol: Hero or Villain?" https://extension.psu.edu/fuel-ethanol-hero-or-villain

